Flash Freezer Temperature Guide: Optimal Settings for 2026
Your flash freezer temperature isn’t just a setting—it’s the decisive factor determining whether your products maintain premium quality or suffer irreversible cellular damage. When water-containing materials freeze slowly at temperatures just below 0°C, large ice crystals form that rupture cell walls, degrade texture, and destroy nutritional value. Flash freezing reverses this destructive process by achieving specific temperature thresholds that transform liquid water to solid so rapidly that microscopic ice crystals form instead, preserving structural integrity across everything from delicate fruits to biological specimens. Understanding these critical temperature requirements helps you select the right equipment and process parameters to maximize quality preservation while managing operational costs.
The science behind effective flash freezing centers on the relationship between cooling rate and ice crystal formation. At conventional freezing temperatures, water molecules gather at relatively few nucleation sites, creating large crystals that physically damage cellular structures. Flash freezing achieves temperatures low enough to activate numerous nucleation sites simultaneously, creating a dense field of microscopic crystals that cause minimal disruption. This temperature-dependent transformation explains why getting your flash freezer temperature right isn’t merely important—it’s the defining factor that separates premium preserved products from compromised inventory.
Liquid Nitrogen Flash Freezing at −196 °C: The Ultimate Cryogenic Temperature
Liquid nitrogen represents the gold standard for achieving the fastest possible freezing speeds, operating at temperatures approaching −196 °C (−320.8 °F) that extract heat so rapidly ice crystal formation occurs almost instantaneously. When products pass through liquid nitrogen systems, the extreme temperature differential creates such intense heat removal that surface layers freeze completely within seconds while maintaining interior quality. This makes liquid nitrogen the preferred choice for applications where absolute maximum freezing speed is required, such as preserving sensitive biological samples or premium food products.
Direct submersion vs. vapor exposure: Which method works best?
Direct immersion in liquid nitrogen produces the fastest possible freezing rates but may cause thermal shock damage to delicate products like thin fish fillets or biological tissues. Controlled exposure to liquid nitrogen vapor typically provides better results for sensitive materials, achieving rapid freezing while reducing surface damage risk. For robust products like thick meat cuts or packaged vegetables, direct submersion often delivers superior results without quality compromise. Always test both methods with your specific product to determine which delivers optimal cellular preservation.
Why liquid nitrogen outperforms mechanical systems for fastest freezing
The extreme cold of liquid nitrogen creates heat transfer rates impossible for even the most advanced mechanical systems to match. While mechanical flash freezers typically operate between −25 °C and −40 °C, liquid nitrogen’s −196 °C temperature provides more than four times the thermal driving force for heat extraction. This translates to freezing times measured in seconds rather than minutes, making liquid nitrogen essential for products where even brief exposure to intermediate temperatures causes quality degradation.
Why −48 °C Represents the Absolute Freezing Threshold for Water

The −48 °C (−54 °F) temperature threshold represents a critical scientific boundary in flash freezing operations—the absolute limit for supercooled water. Below this temperature, water’s molecular structure physically transforms into tetrahedron shapes that force crystallization regardless of purity or container conditions. Understanding this threshold is essential because it defines the minimum temperature required to guarantee complete solidification without relying on unpredictable nucleation events.
Understanding the science behind supercooled water’s limit
Supercooling occurs when extremely pure water in smooth containers remains liquid far below 0°C due to lack of nucleation sites. This metastable state can persist until disturbed or cooled sufficiently, at which point crystallization happens within nanoseconds. Commercial flash freezing processes typically operate well below the −48 °C threshold to ensure consistent results regardless of minor variations in product characteristics or container surfaces that might otherwise trigger unpredictable freezing behavior.
How to prevent supercooling failures in your flash freezing process
Temperature fluctuations during freezing can dramatically affect ice crystal formation and final quality. Even brief warming periods that don’t fully thaw products allow existing ice crystals to grow larger, partially negating flash freezing benefits. Implement rigorous temperature monitoring throughout your freezing cycle, especially through the critical −1 °C to −5 °C zone where ice crystal formation occurs. Modern systems with multiple temperature probes and data logging ensure consistent results by validating that all products achieve required cooling profiles.
Mechanical Flash Freezer Temperature Range: −25 °C to −40 °C Explained
Commercial mechanical flash freezers operate at temperatures significantly warmer than liquid nitrogen systems but have been engineered to achieve freezing rates sufficient for most food preservation applications. The typical operating range of −25 °C to −40 °C (−13 °F to −40 °F) provides enough cooling capacity when combined with optimized airflow or direct-contact cooling surfaces. Higher-moisture and denser products generally require temperatures at the lower end of this range to achieve adequate freezing rates.
Air impingement systems: Achieving maximum freezing rates at higher temperatures
Advanced airflow designs that direct high-velocity jets of cold air across product surfaces achieve heat transfer coefficients far exceeding conventional forced-air systems. These air impingement systems allow you to maintain slightly warmer freezer temperatures while still achieving rapid freezing, reducing energy consumption without sacrificing quality. For example, an air impingement system operating at −30 °C often achieves better freezing rates than a conventional system at −35 °C, providing significant energy savings over time.
How product density affects your optimal mechanical flash freezing temperature
Thicker, denser products require lower temperatures and longer exposure times to achieve proper flash freezing. A 1-inch thick fish fillet might freeze adequately at −30 °C, while a 3-inch thick steak requires −38 °C to achieve the same rapid freezing rate through the entire product mass. Pro tip: Always validate your specific product’s freezing profile using center-temperature probes rather than relying solely on chamber temperature readings, as the difference between chamber temperature and actual product temperature can create quality inconsistencies.
Food Industry Flash Freezing Temperatures: Optimal Ranges by Product Type
Commercial food flash freezing typically targets temperatures between −25 °C and −40 °C to achieve rapid freezing rates necessary for optimal quality preservation. The primary goal is passing through the critical temperature zone (−1 °C to −5 °C) as quickly as possible to minimize cellular damage and preserve texture, flavor, and nutritional value. Different food categories require specific temperature optimization based on their unique composition and structure.
Fruits and vegetables: Why lower temperatures prevent mushy texture
High-moisture produce like berries, melons, and leafy greens demands the lowest mechanical flash freezer temperatures (−35 °C to −40 °C) to prevent the cellular damage causing mushy texture after thawing. The rapid freezing creates microscopic ice crystals that don’t rupture cell walls, preserving crisp texture and vibrant color. Critical mistake to avoid: Freezing delicate fruits at temperatures above −30 °C often results in unacceptable texture degradation despite appearing frozen.
Meats and seafood: Temperature requirements for maximum juiciness retention
Fatty fish and meats have different freezing characteristics based on lipid composition, requiring temperature profiles optimized for specific properties. Fatty fish like salmon benefit from slightly warmer temperatures (−28 °C to −32 °C) to prevent lipid crystallization issues, while lean meats like chicken breast require colder temperatures (−35 °C to −40 °C) to maintain juiciness. Consistently passing through the critical zone in under 30 minutes preserves the cellular structure that retains natural juices during cooking.
Biological Sample Preservation: Critical Temperature Requirements for Viability
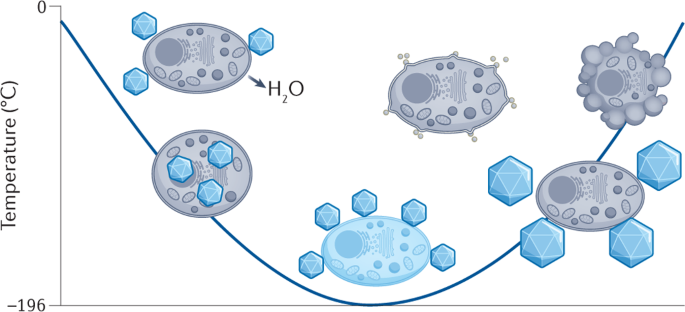
Biological samples demand extremely low temperatures to prevent cellular damage during storage, with flash freezing typically achieved through submersion in liquid nitrogen (−196 °C) or dry ice/ethanol mixtures (−78.5 °C). These temperatures essentially halt all biological and chemical processes, allowing indefinite storage while preserving the sample’s original structure and viability. Living cells are far more sensitive to ice crystal damage than food products, making precise temperature control absolutely critical.
−80 °C vs. −196 °C: Choosing the right temperature for your samples
While liquid nitrogen storage at −196 °C provides ultimate protection against degradation, many biological samples remain viable at −80 °C, which is achievable with specialized mechanical freezers. Tissue samples and cell cultures often maintain viability at −80 °C for years, while more sensitive specimens like stem cells or certain enzymes require liquid nitrogen temperatures. Expert note: Always verify your specific sample type’s temperature requirements through literature review or pilot testing before committing to large-scale preservation.
How to prevent cellular damage during biological flash freezing
The scientific rationale for biological flash freezing focuses on minimizing ice crystal size to prevent mechanical damage to cell membranes and organelles. When samples freeze slowly, large ice crystals form both inside and outside cells, often proving fatal. Flash freezing creates numerous small crystals causing minimal disruption, significantly improving survival rates after thawing. For maximum viability, ensure your flash freezing process achieves cooling rates exceeding 10°C per minute through the critical temperature zone.
Storage Temperature Requirements After Flash Freezing: What Happens Next
The temperature requirements for storing flash-frozen products differ significantly from those needed for initial freezing, though both are critical for maintaining quality. After flash freezing, products must be stored at temperatures low enough to prevent ice crystal growth during storage. Most food products require storage temperatures of −18 °C to −24 °C for extended shelf life, while biological samples often need −80 °C or lower to prevent degradation.
Why −18 °C to −24 °C works for most food products
These storage temperatures maintain ice crystals in their small, flash-frozen state without allowing growth that would damage cellular structures. Consistent storage below −18 °C prevents recrystallization—the process where small ice crystals merge into larger damaging ones during temperature fluctuations. Warning: Even brief temperature excursions above −12 °C can trigger significant recrystallization, making reliable freezer performance and monitoring essential for quality maintenance.
Temperature excursions: The hidden danger to flash-frozen quality
Partial thawing and refreezing creates larger ice crystals causing the cellular damage flash freezing was designed to prevent. Storage systems must maintain consistent temperatures without interruption, requiring reliable refrigeration equipment and backup systems for critical applications. Implement temperature monitoring with alarms that alert staff to excursions before they cause significant product damage—this simple step prevents millions in annual losses for commercial operations.